【讲座主题】Ammonia as Chemical Energy Carrier: Electro-Catalytic Synthesis and Decomposition
【讲座时间】2026年3月26日 14:00-15:00
【讲座地点】主楼D260
【主讲人】Olaf Deutschmann 教授
【主讲人简介】Olaf Deutschmann,德国卡尔斯鲁厄理工学院(KIT)化工技术讲席教授,化学技术与聚合物化学研究所主任,国际燃烧学会会士。1996年于海德堡大学化学系获得博士学位,2001年获得海德堡大学物理化学任教资格。曾先后在海德堡大学、美国明尼苏达大学、洛斯阿拉莫斯国家实验室及卡尔斯鲁厄大学工作,2006年至今任职于卡尔斯鲁厄理工学院。研究聚焦环境友好和气候友好的新型化工技术,涵盖无碳化学储能载体、燃料电池与电解池、反应工程、非均相催化、材料合成及多相流等方向。创建多相化学反应流模拟软件DETCHEM,获Max Buchner研究基金大奖。已发表SCI论文370余篇,被引超17500次,受邀作学术报告和讲座100余次。
【讲座内容】
Ammonia as Chemical Energy Carrier: Electro-Catalytic Synthesis and Decomposition
Olaf Deutschmann, Karlsruhe Institute of Technology (KIT),Germany
deutschmann@kit.edu
The talk focuses on alternative electro- and thermo-catalytic pathways for the production and decomposition of ammonia related to its use as carrier of renewable energy. Recent advances in electrocatalytic ammonia synthesis in proton-conducting ceramic cells (PCCs) are discussed with a focus on iron- and ruthenium- based electrodes [1]. The effects of temperature, gas flow, voltage, and electrolyte thickness on electrochemical ammonia synthesis are investigated. To differentiate the various effects and mechanisms contributing to the electrocatalytic formation of NH3, different gas flow configurations are studied. The experimental results demonstrate that NH3 formation is primarily governed by the applied cell voltage, while the current density plays only a minor role. A strong interaction between electro- and thermo-catalytic reactions occurs.
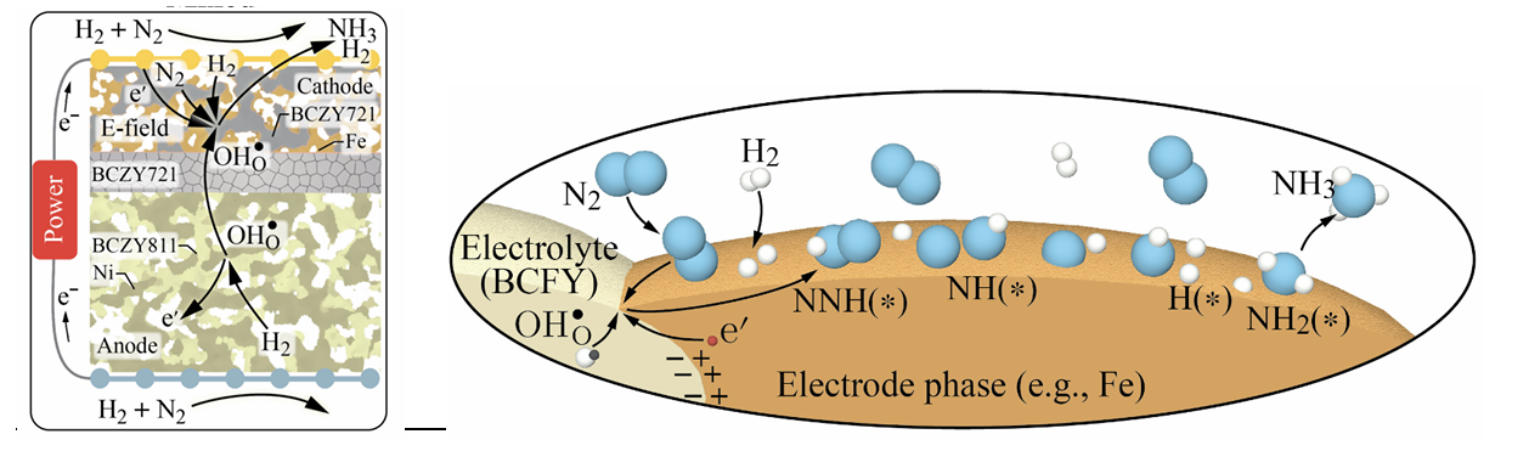
Figure 1: Schematic representation of reactions occurring at a proton-conducting ceramic cell that either contribute to NH3 synthesis or compete with it. The reactions take place simultaneously; adapted from [1].
Co-feeding H2 at the cathode proved advantageous for optimizing reaction conditions and increasing ammonia synthesis rates to values of 3×10−8 mol s−1 cm−2 using a PCC with an iron based electrode of an active area of 12.57 cm²; both values are much higher than reported in literature before.
Also, decomposition of NH3 to H2 can be conducted in PCC. In all cases both electro- and thermos-catalytic processes have to be understood [2]. The combination of thermo-catalytic and electrochemically supported ammonia synthesis and decomposition opens new pathways for the electrification of NH3 production using the rather inexpensive material iron and the use of NH3 as hydrogen carrier.
[1] P. Blanck, E.P. Martin, D. Schmider, J. Dailly, R.J. Kee, O. Deutschmann. Electrochemical Ammonia Synthesis in a Proton-conducting Ceramic Cell: A Parameter Study of an Iron-based Electrode. J. Electrochem. Soc. (2025) DOI: 10.1149/1945-7111/adfc9e.
[2] S. Davari, R. Chacko, T. Bastek, P. Lott, J. Dailly, S. Angeli, O. Deutschmann. Experimental and Microkinetic Investigation of Thermo-Catalytic Ammonia Decomposition over a Ba-promoted Ru/Ni BCZY Catalyst for Use in Ammonia-fed Protonic Ceramic Cells. Appl. Catal. A. 708 (2025) 120571. DOI:10.1016/j.apcata.2025.120571.
